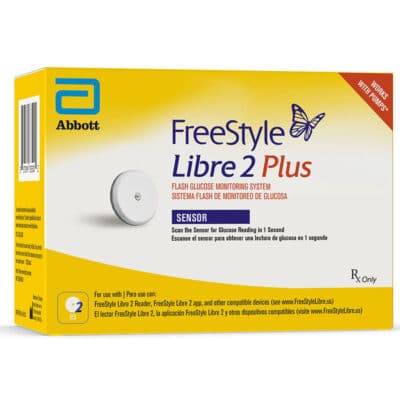
Abbott® FreeStyle Libre 2 Plus Sensor
If you’ve used Abbott®’s FreeStyle Libre 2 System in the past, you already know how reliable* this continuous glucose monitoring (CGM) system’s sensors can be. The FreeStyle Libre 2 Plus sensor combines that peace of mind with new features like a 15-day lifespan and Tandem® t:slim X2 insulin pump compatibility. Getting your hands on one of these sensors shouldn’t be difficult—just place an order from Advanced Diabetes Supply.
Please call our Customer Care Team at 1-866-422-4866 or fill out the form below to see if you qualify for a CGM sensor with $0 out-of-pocket costs.†
With the FreeStyle Libre 2 Plus sensor, you’ll find it easier than ever to keep track of your glucose levels. This device takes the fingerstick-free** glucose testing you know and love from the original FreeStyle Libre 2 sensor and brings this functionality to the next level. Among other benefits, these sensors boast an improved MARD¶ and a 15-day service life.
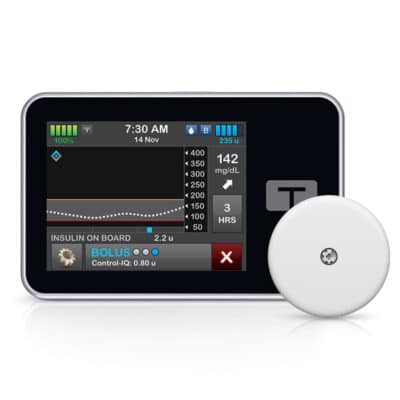
Furthermore, the FreeStyle Libre 2 Plus sensor is compatible with the Tandem t:slim X2 insulin pump. By using these products together, diabetes patients will have an opportunity to create a hybrid closed-loop system.
- First 15-day CGM sensor in the U.S.
- Longest-lasting sensor
- The same ease of use as the “standard” FreeStyle Libre 2‡
- Improved accuracy and an enhanced MARD of 8.2 percent¶
- Readings every minute†
- No user calibration
- Compatibility with automated insulin dosing systems like the t:slim X2¶
- Works with the FreeStyle Libre 2 app§, LibreView||, and LibreLinkUp#
- Can be worn by people with diabetes age two years and older
The FreeStyle Libre 2 Plus sensor is a new-and-improved iteration of the sensors found in Abbott’s FreeStyle Libre 2 System. Thus, these devices have the same responsibility as any other CGM sensor—they can measure a wearer’s glucose levels without fingerstick testing**.
If you already use the FreeStyle Libre 2 System but are interested in the possibility of switching to a hybrid closed-loop system for insulin delivery, upgrading to FreeStyle Libre 2 Plus sensors can help. This product is also an excellent fit for t:slim X2 users who would like to establish a hybrid closed-loop system based on a FreeStyle Libre-brand CGM.
For people who do not already use the FreeStyle Libre 2 System, FreeStyle Libre 2 Plus sensors are available alongside the reader for this system. Alternatively, current FreeStyle Libre 2 System users can order the updated sensors for use with their existing equipment. (FreeStyle Libre 2 Plus sensors are not compatible with the reader or app used in the FreeStyle Libre 3 System.)
Before using your FreeStyle Libre 2 Plus sensor, take a close look at this product’s official user guide.
Important Safety Information:
RX ONLY. The t:slim X2 pump and Control-IQ technology are intended for single patient use. The t:slim X2 pump and Control-IQ technology are indicated for use with NovoLog or Humalog U-100 insulin. t:slim X2 insulin pump: The t:slim X2 insulin pump with interoperable technology is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in people requiring insulin. The pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices. The pump is indicated for use in individuals 6 years of age and greater. Control-IQ technology: Control-IQ technology is intended for use with compatible integrated continuous glucose monitors (iCGM, sold separately) and alternate controller enabled (ACE) pumps to automatically increase, decrease, and suspend delivery of basal insulin based on iCGM readings and predicted glucose values. It can also deliver correction boluses when the glucose value is predicted to exceed a predefined threshold. Control-IQ technology is intended for the management of Type 1 diabetes mellitus in persons 6 years of age and greater.
WARNING:
Control-IQ technology should not be used by anyone under the age of 6 years old. It should also not be used in patients who require less than 10 units of insulin per day or who weigh less than 55 pounds.
How Many Times Can You Scan a FreeStyle Libre 2 Plus Sensor?
There is no limitation on the number of times a FreeStyle Libre 2 Plus user can safely scan their sensor.
Does the Libre 2 Plus Sensor Leave a Needle in Your Arm?
While FreeStyle Libre 2 Plus sensors rely on a needle as part of their application process, it does not stay in the skin after application is complete. Instead, the needle creates a small puncture to get a sterile filament under a wearer’s skin—while the filament stays behind, the needle comes out immediately.
How Often Does the Libre 2 Plus Sensor Check Glucose?
The FreeStyle Libre 2 Plus sensor takes updated glucose readings once per minute. These readings are then stored for up to eight hours in 15-minute intervals.
Does Tylenol Interfere With FreeStyle Libre 2 Plus?
Acetaminophen (AKA Tylenol) should not interfere with your FreeStyle Libre 2 Plus sensor.
What Is the Wear Time for the FreeStyle Libre 2 Plus?
Unlike previous Abbott CGM sensors, FreeStyle Libre 2 Plus sensors can be worn for up to 15 days.
† FreeStyle Libre 2 Plus sensor connects via Bluetooth to the Tandem t:slim X2 insulin pump. Glucose data is visualized on the t:connect mobile app and t:slim X2 insulin pump every minute.
*High levels of vitamin C may affect the accuracy of sensor readings. Refer to the FreeStyle Libre 2 user manual for more information.
†For qualified persons with applicable in-network insurance and subject to CGM coverage requirements. Deductibles, co-pays, and other conditions apply. Speak to an ADS representative and your insurance company to confirm your coverage and costs.
‡Haak, T. Diabetes Therapy (2017): https://doi.org/10.1007/s13300-016-0223-6.
¶FreeStyle Libre 2 User’s Manual
- The FreeStyle Libre 2 app is only compatible with certain mobile devices and operating systems. Please check the website for more information about device compatibility before using the app. Use of FreeStyle Libre 2 app requires registration with LibreView.
||The LibreView data management software is intended for use by both patients and healthcare professionals to assist people with diabetes and their healthcare professionals in the review, analysis and evaluation of historical glucose meter data to support effective diabetes management. The LibreView software is not intended to provide treatment decisions or to be used as a substitute for professional healthcare advice.
#The LibreLinkUp app is only compatible with certain mobile device and operating systems. Please check www.librelinkup.com for more information about device compatibility before using the app. Use of the LibreLinkUp app requires registration with LibreView. LibreLinkUp is not intended to be used for dosing decisions. The user should follow instructions on the continuous glucose monitoring system. LibreLinkUp is not intended to replace self-monitoring practices as advised by a physician.
**Fingersticks are required if your glucose alarms and readings do not match symptoms or when you see Check Blood Glucose symbol during the first 12 hours.